
What Do Cancer Cells Eat?
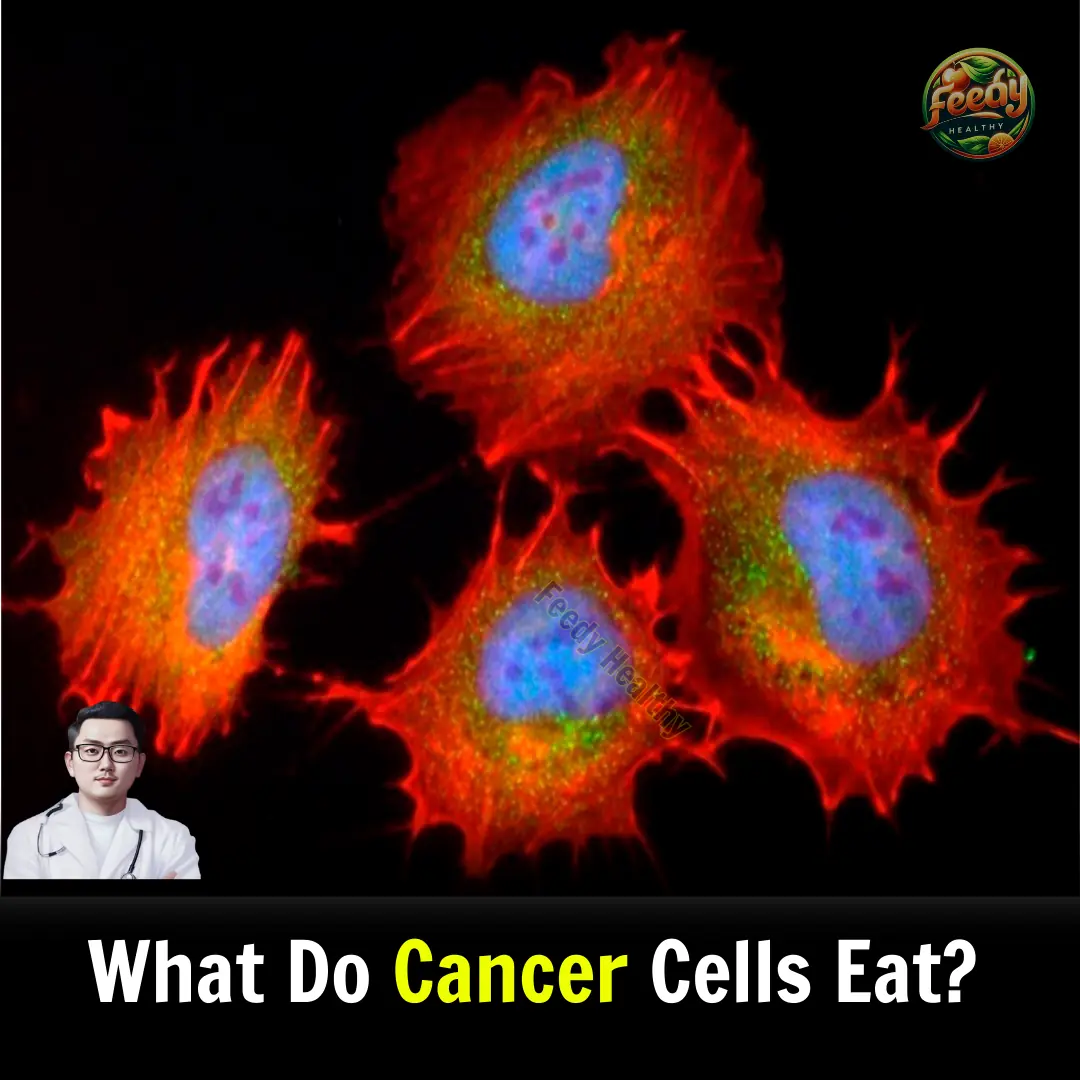
Cancer cells primarily feed on glucose, the same type of sugar consumed by healthy cells. However, cancer cells consume 3 to 4 times more glucose than normal cells to meet their high energy and biosynthesis demands. Understanding how these cells metabolize energy is crucial for developing more effective cancer treatments.
Unlike normal cells, which optimize energy production through mitochondrial respiration, tumor cells adopt unique strategies to support their growth. This article explores the energy sources that fuel cancer cells and how they relate to tumor progression and targeted therapies.
The Warburg Effect: Preference for Aerobic Glycolysis
One of the most striking features of cancer cell metabolism is the Warburg Effect, first described by Otto Warburg in 1924. This phenomenon refers to the tendency of tumor cells to use aerobic glycolysis—converting glucose into lactate even in the presence of oxygen—rather than mitochondrial oxidative phosphorylation.
Under normal conditions, healthy cells metabolize glucose via mitochondrial respiration, generating up to 36 ATP molecules per glucose. In contrast, cancer cells rely mainly on glycolysis, producing only 2 ATPs per glucose, but at a much faster rate.
This adaptation offers several advantages for cancer:
-
Production of metabolic intermediates: Glycolysis intermediates are diverted into anabolic pathways like nucleotide and lipid synthesis, essential for cell proliferation.
-
Tolerance to low oxygen levels: In oxygen-poor tumor regions, glycolysis ensures continued energy production.
-
Evasion of regulation mechanisms: Prioritizing glycolysis alters mitochondrial regulation, enhancing cancer cell survival.
Moreover, the Warburg effect is not limited to individual tumor cells—it also reshapes the tumor microenvironment. This metabolically altered setting creates a supportive ecosystem for cancer progression, where cells exchange metabolites like lactate, which can be reused by neighboring tumor cells for energy or biosynthesis.
Alternative Energy Sources in Cancer Cells
Although glucose is the primary fuel, cancer cells also take up other molecules to fulfill their metabolic needs:
Glutamine
Glutamine is the second most important fuel for tumors. This amino acid is converted into glutamate by glutaminase, which then enters the Krebs cycle as α-ketoglutarate. Beyond energy, glutamine supports nucleotide, lipid, and glutathione synthesis—a key antioxidant maintaining redox balance.
Recent studies show that certain cancers rely heavily on glutamine metabolism, making it a promising target for therapy.
Lipids
Lipids serve not only as structural components of cell membranes but also as an energy source through β-oxidation of fatty acids. In many tumors, lipid metabolism is dysregulated, increasing fatty acid availability to fuel cell metabolism.
Additionally, cancer cells can produce lipids de novo—from scratch—to generate phospholipids necessary for membrane expansion during cell division.
Other Amino Acids
Cancer cells also consume amino acids like serine and glycine, which play roles in carbon metabolism and macromolecule synthesis. These molecules contribute to NADPH production, essential for managing oxidative stress.
For example:
-
Serine is vital for purine and pyrimidine synthesis.
-
Glycine is crucial for collagen production and structural protein synthesis.
Comparing Metabolism: Normal Cells vs. Cancer Cells
The metabolism of cancer cells is markedly different from that of healthy cells. While normal cells efficiently generate energy via mitochondrial respiration, tumor cells prioritize speed over efficiency.
They rewire their metabolism to meet increased biosynthetic demands, using multiple energy sources and adapting to harsh conditions like low oxygen or nutrient deprivation.
One striking feature is their metabolic flexibility: cancer cells can switch between energy sources depending on their environment. For example, under severe oxygen deprivation, they increase their reliance on glutamine or tap into stored fatty acid breakdown.
Therapeutic Advances Based on Cancer Metabolism
Understanding cancer cell metabolism and the fuels that support their growth has led to promising therapies targeting these vulnerabilities. Some of the most promising strategies include:
Metabolic Inhibitors
Drugs targeting key enzymes like glutaminase and hexokinase aim to disrupt essential metabolic pathways. For instance, glutaminase inhibitors reduce the glutamine supply to the Krebs cycle, impairing cell proliferation. Similarly, PDK inhibitors (pyruvate dehydrogenase kinase) may reactivate mitochondrial respiration in tumor cells.
Dietary Strategies
Ketogenic diets and calorie restriction have shown promise in preclinical studies by lowering glucose availability, forcing cancer cells to depend on less efficient energy sources. Intermittent fasting may also reduce insulin levels and enhance chemotherapy effectiveness.
Combination Therapies
Combining metabolic inhibitors with traditional treatments like chemotherapy and radiation can enhance therapeutic outcomes by exploiting cancer metabolism vulnerabilities. This multifaceted approach targets both cell growth and metabolic adaptability.
Conclusion
Cancer cell metabolism is a dynamic and essential area of research for developing new therapies. By understanding how tumors derive energy, researchers can design more effective strategies to halt their growth and spread.
Targeted therapies that exploit cancer’s unique metabolic traits represent a promising direction for more effective and personalized treatments. Moreover, this knowledge supports combining traditional approaches with new metabolic strategies, expanding our arsenal against cancer.
Let me know if you’d like this in a more academic tone or formatted for a social media post, blog article, or infographic!
News in the same category


If Your Kidneys Are in Danger, Your Body Will Send You These 8 Signals — Don’t Ignore Them

The Surprising Effects of Avocado on Your Heart and Brain
Natural Remedy for Cataracts and Eye Inflammation: Restore Your Vision Naturally

Unlock the Golden Magic of Corn Silk Tea
9 Powerful Home Remedies to Get Rid of Fungal Infection (Daad, Khaj, Khujli) Fast
7 Shocking Health Benefits Of Eating Sweet Potatoes Every Day — According To Science

About 15 Minutes Before a Stroke, the Body Often Sends 4 Clear Warning Signs — Call Your Loved Ones Immediately
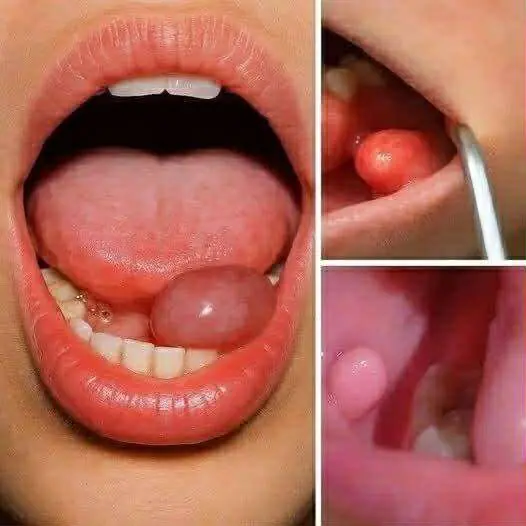
Hidden Dangers in Your Mouth: Early Signs of Oral Cancer

The Secret Power Of The Herb That Helps You Age Gracefully

The Unexpected Benefits of Eating Chicken Feet
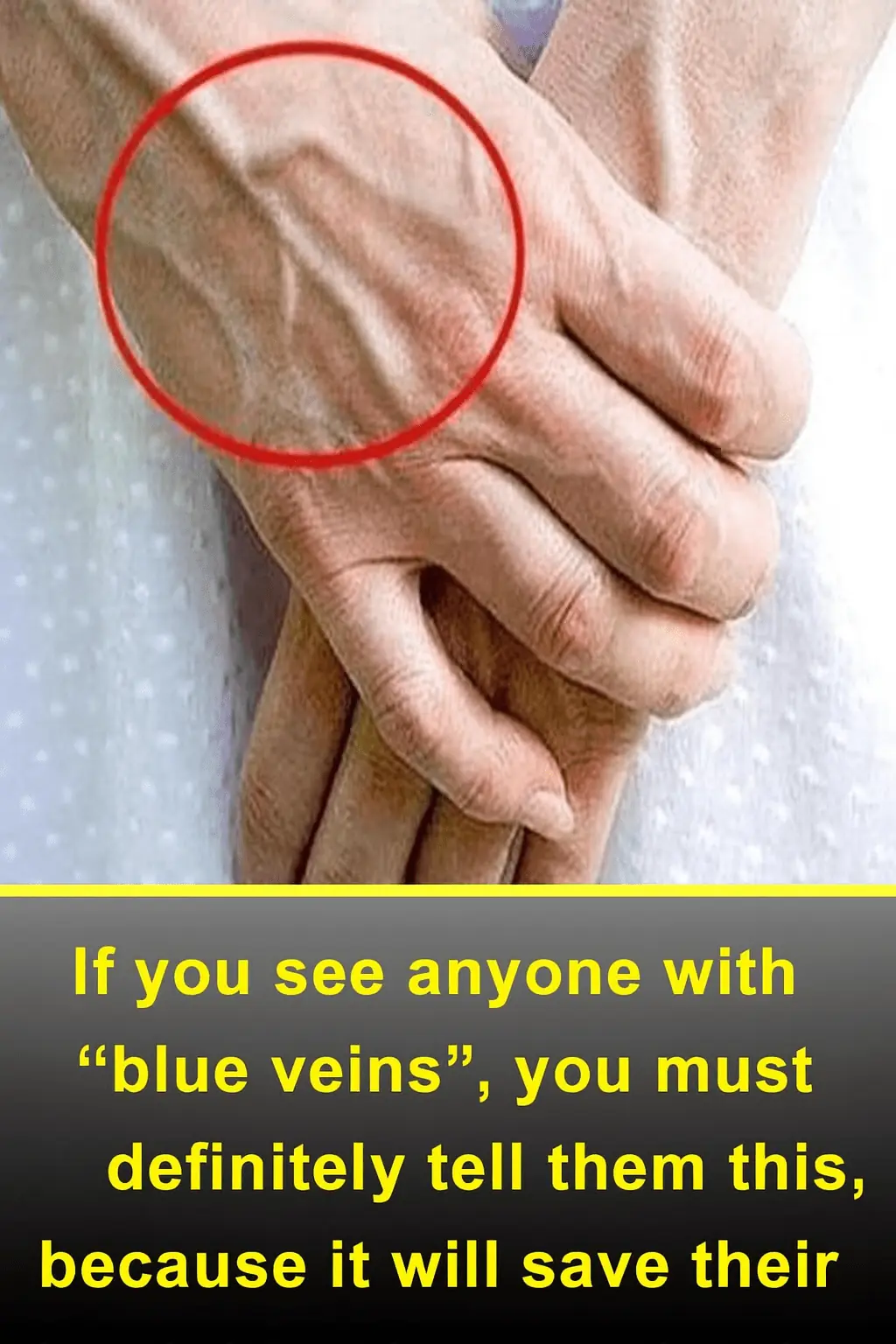
If You See Someone with “Blue Veins,” Tell Them This — It Could Save Their Life
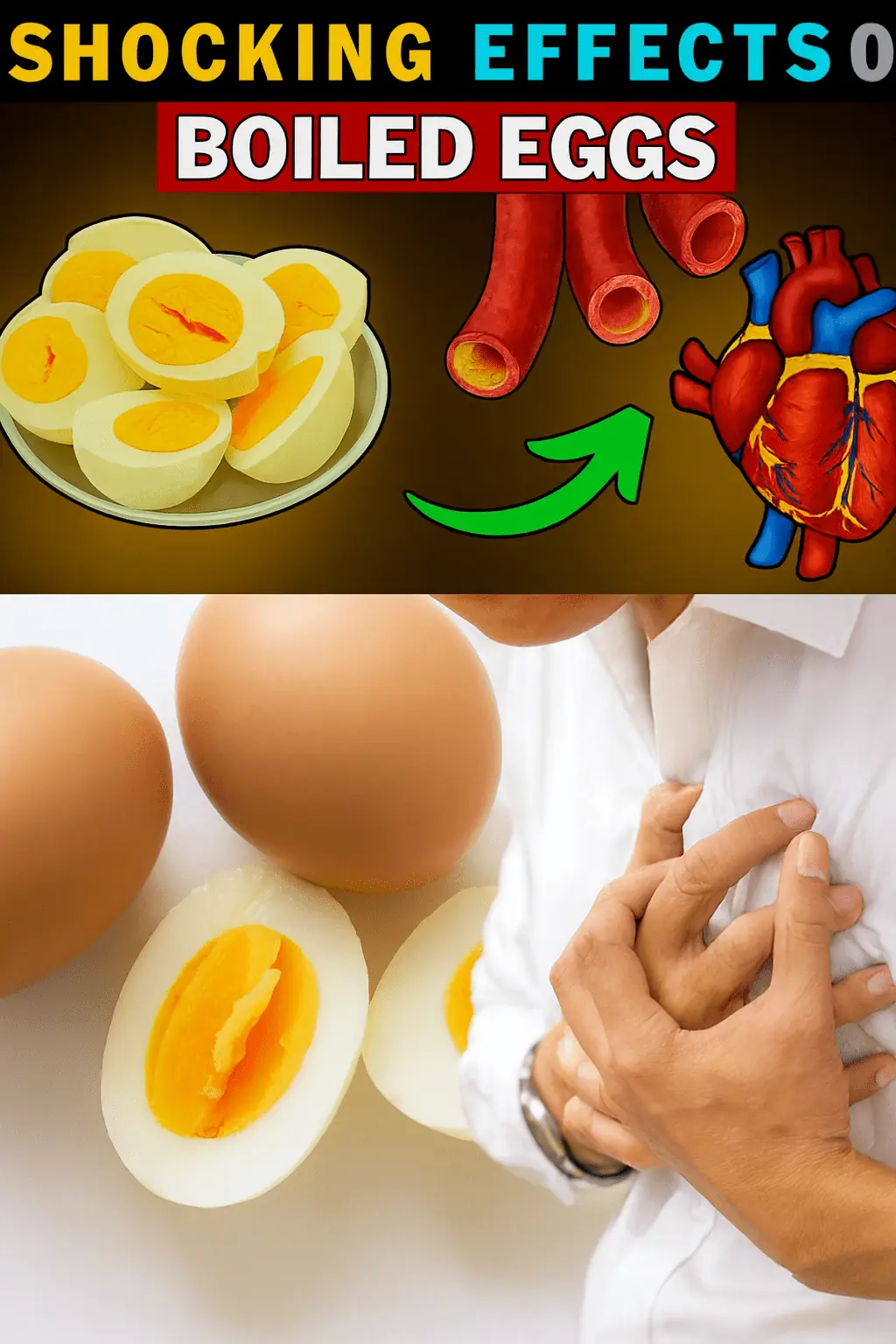
The Secret Power of Two Eggs a Day: Could This Simple Habit Transform Your Health? Buy vitamins and supplements

Man Passed Away After Eating Eggs — Stop Eating Eggs This Way Immediately
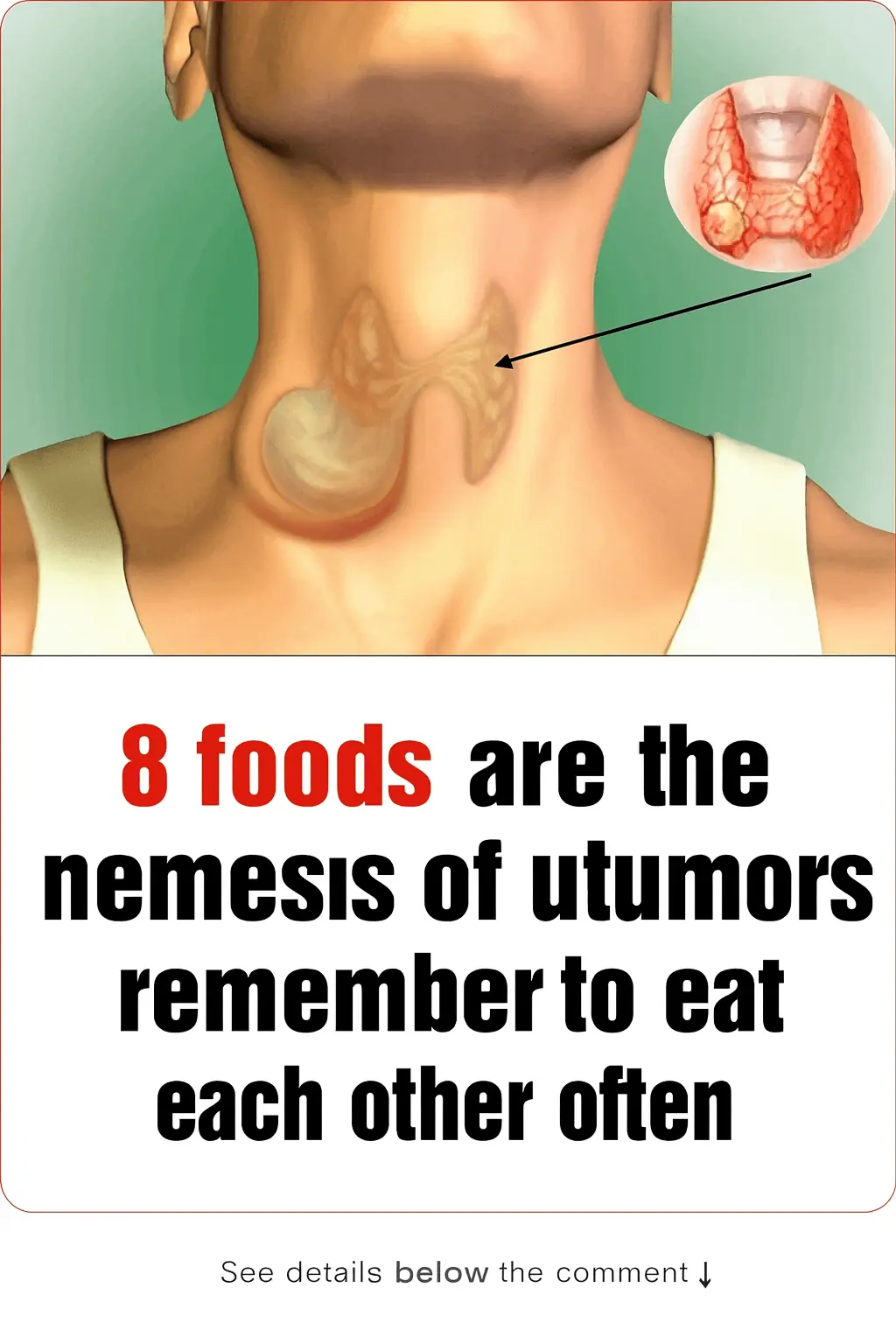
8 Foods That Fight Tumors — Eat Them Regularly

Does Eating Bananas Before Bed Have Any Benefits?

The Tongue as a Health Indicator: Meaning of a Whitish Color
5 early warning signs of cervical cancer
7 Innocent Mistakes That Get Your Kidneys in Big Trouble
News Post
WHAT HAPPENS WHEN WE TONGUE KISS…See more

Nature’s Secret: 4 Healing Leaves That Support Metabolism, Immunity & Circulation Naturally

Don’t Drink Coconut Water Before You Know These 11 Secrets!

Pumpkin Seed Milk — The Natural Parasite Cleanser
Fast Rice Water Trick for a Brighter Smile
Morning Drink to Revive Your Kidneys Fast
The Onion Recipe That Could Transform Your Blood Sugar, Support Cleaner Arteries, and Protect Your Heart!

Top 4 Fruits That Help Your Kidneys Flush Out Toxins While You Sleep
Ginger, Clove, and Honey: The Natural Trio Your Body Will Thank You For

Heal 15 Years of Joint Pain Naturally with Turmeric and Honey Tea

This Juice Revived My Grandma’s Energy — Say Goodbye to Fatigue and Body Pain with This Natural Recipe

The Benefits of Eating 2 Boiled Eggs Every Morning: Transform Your Health!

If Your Kidneys Are in Danger, Your Body Will Send You These 8 Signals — Don’t Ignore Them

The Surprising Effects of Avocado on Your Heart and Brain

Ways to Get Over a Man Who Didn’t Value You

I’m 66 but Look 36 — My Secret? Aloe Vera & Ginger for Firm, Smooth Skin

How to Make Okra Water to Treat 17 Health Problems Naturally

Banana and Egg Mask to Look Younger Even in Your 80s

Scent Leaf Secrets Unveiled: 10 Surprising Health Benefits of This Miracle Herb
