
UCLA Scientists Unveil PP405 Molecule as Potential Breakthrough in Hair Regrowth
Los Angeles, CA — Scientists at the University of California, Los Angeles (UCLA) have announced encouraging results from early-stage clinical trials of a molecule known as PP405, a compound that appears to awaken dormant hair follicle stem cells and restore natural hair growth.
Unlike current treatments such as minoxidil or finasteride, which primarily slow hair loss, PP405 works by directly reactivating the biological machinery of hair follicles. This regenerative approach has raised hopes for what some researchers are calling the closest prospect yet of a genuine cure for baldness.
Early Clinical Findings
The molecule was initially identified during research into cellular metabolism and stem cell biology. By blocking the mitochondrial pyruvate carrier (MPC), PP405 alters how hair follicle cells use energy, effectively “switching on” follicles that had entered long-term dormancy.
In Phase 1 clinical trials conducted at UCLA in 2023, researchers confirmed that PP405 could be applied topically without entering the bloodstream and without triggering serious side effects. Within just one week of testing, hair follicle activation was observed under clinical monitoring.
More recently, Phase 2a trials conducted in mid-2025 involved over 78 male and female participants with androgenetic alopecia (pattern baldness). Results showed that after eight weeks of application, nearly one-third of participants experienced over 20% improvement in hair density. Importantly, the treatment was well tolerated, with no systemic absorption detected.
A Global Health Impact
Baldness is not only a cosmetic concern but also a widespread medical condition. Studies suggest that more than 50% of men and around 25% of women will experience significant hair loss by age 50. Current solutions, ranging from pharmaceutical lotions to surgical hair transplants, offer only temporary or partial relief.
If PP405 continues to demonstrate safety and effectiveness in larger-scale trials, it could mark a revolution in hair restoration, offering millions a chance at regaining thicker, healthier hair without invasive procedures.
The Road Ahead
Despite the promising data, experts caution that PP405 is still years away from market availability. Larger Phase 3 clinical trials, expected to begin in 2026, will be critical in confirming long-term safety and efficacy across diverse patient groups. Regulatory approval from the U.S. Food and Drug Administration (FDA) and global agencies would follow only if results remain positive.
Dr. William Lowry, a stem cell biologist at UCLA involved in the original discovery, emphasized the broader implications:
“PP405 is not just about hair. It highlights how regenerative medicine can restore tissues once thought permanently damaged or inactive.”
A Glimpse of Hope
For now, PP405 represents a beacon of hope in the long and often frustrating battle against baldness. Should ongoing trials succeed, the molecule may not only transform the billion-dollar hair loss industry but also reshape public attitudes toward aging and regenerative therapies.
As one commentator put it in a recent medical review: “This is the first time we are talking seriously about a potential cure, not just a management tool, for hair loss.”
News in the same category

🌿 3 Nourishing Drinks to Support Leg Strength, Circulation & Comfort After 60
Warning Signs in Your Eyes: When to Seek Urgent Medical Attention
Flush Out Toxins Naturally: Support Your Kidneys, Liver, and Lungs for Vibrant Health
Garlic and Onion: The Natural Secret to Better Hearing
8 Shocking Signs of Pancreatic Cancer You Shouldn't Ignore

Euphorbia Hirta: The Backyard Weed With Golden Healing Powers

Germany’s Cartilage Repair Gel: Hope, Hype, and the Future of Joint Regeneration

Bioengineered Heart Patch Could Transform Treatment for Heart Failure

No More Fine Lines Thanks To Clove Orange Anti-Aging Oil

Age 40 Is the Decisive Point for Life Expectancy:
12 signs that may signal a brain aneurysm — Don’t ignore them
Homemade Teeth Whitening: White Teeth in Just 2 Minutes!

The surprising truth about eating eggs every day

5 “Dirtiest” Parts of a Pig That Butchers Never Feed Their Families
🦶 Swollen Feet: A Sign You Shouldn’t Ignore — What Your Body Might Be Telling You

🚩 19 Possible Signs of Cancer: When to See a Doctor

4 “Golden” Signs in Your Feet That Reveal Longevity: If You Have Them, You’re Likely to Live a Healthy Life
News Post

A Gentle Herbal Infusion to Support Kidney Health and Natural Detox

🌿 3 Nourishing Drinks to Support Leg Strength, Circulation & Comfort After 60

Warning Signs in Your Eyes: When to Seek Urgent Medical Attention

Flush Out Toxins Naturally: Support Your Kidneys, Liver, and Lungs for Vibrant Health
8 Common Foods That May Harm Prostate Health – And What to Choose Instead

Brazil Deploys AI “Tree-Hugging” Devices to Combat Illegal Logging in the Amazon

Garlic and Onion: The Natural Secret to Better Hearing

8 Shocking Signs of Pancreatic Cancer You Shouldn't Ignore
Onions: The Hidden Superfood Every Man Needs for a Healthier Prostate
Unlock the Secret to Clear Lungs: The Astonishing Power of Onions

Unleash Your Inner Vitality with Gbogbo Nise: Nature’s Ultimate Wellness Elixir 🌿

Patrick Kilonzo Mwalua: The “Waterman of Tsavo” Who Brought Life to Kenya’s Wildlife

Euphorbia Hirta: The Backyard Weed With Golden Healing Powers

🧁 Beach-Themed Vanilla Cupcakes Recipe

Horror Film Verónica Sparks Intense Reactions Among Viewers
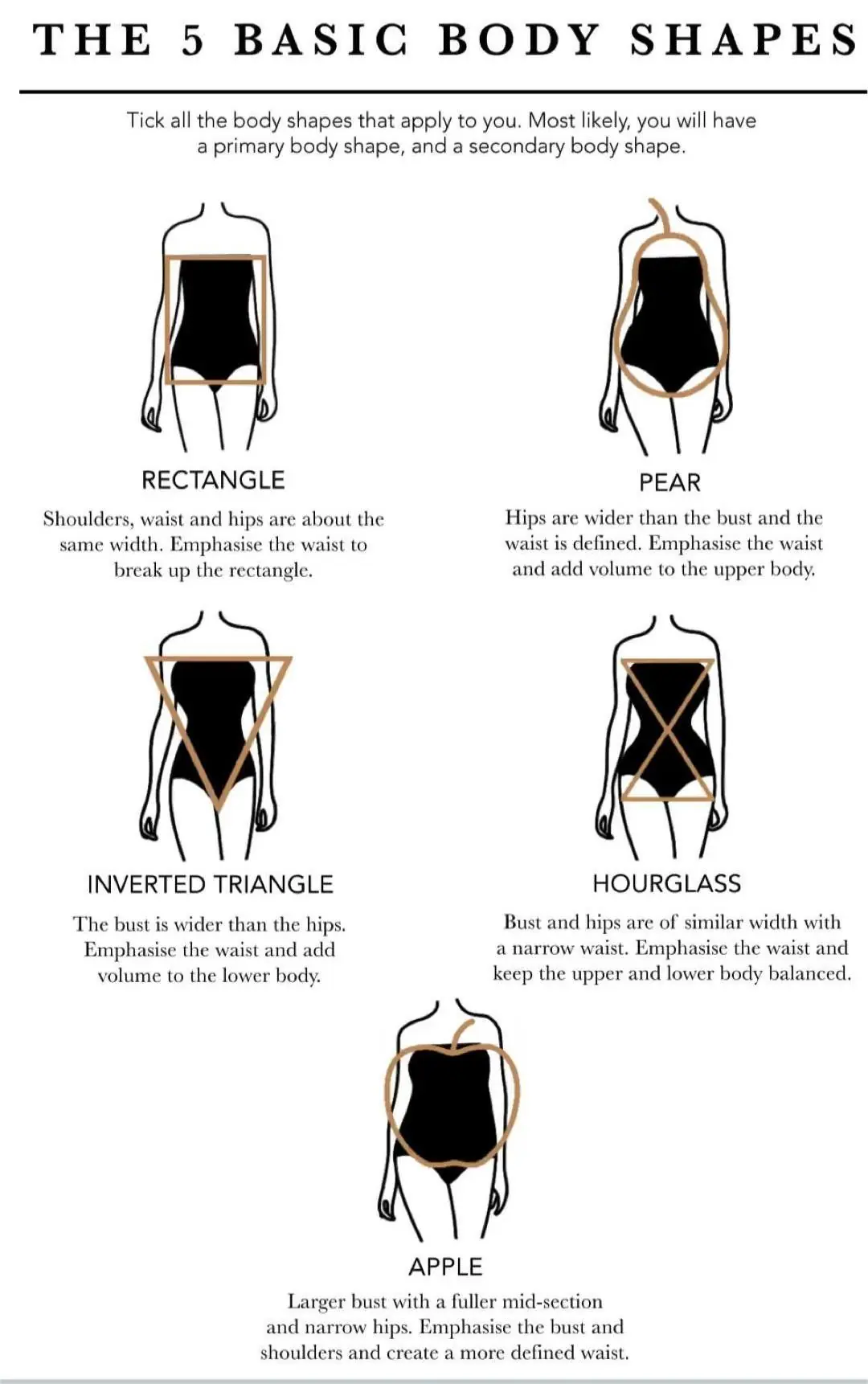
What Body Shape Are You? Easy Steps to Identify It

Germany’s Cartilage Repair Gel: Hope, Hype, and the Future of Joint Regeneration

Bioengineered Heart Patch Could Transform Treatment for Heart Failure

